Project 8
HIV-1 spread in complex cell systems
.
.
.
Bildnachweis:
Universität Heidelberg,
Kommunikation und Marketing
.
.
.
Summary Project description 2nd funding period
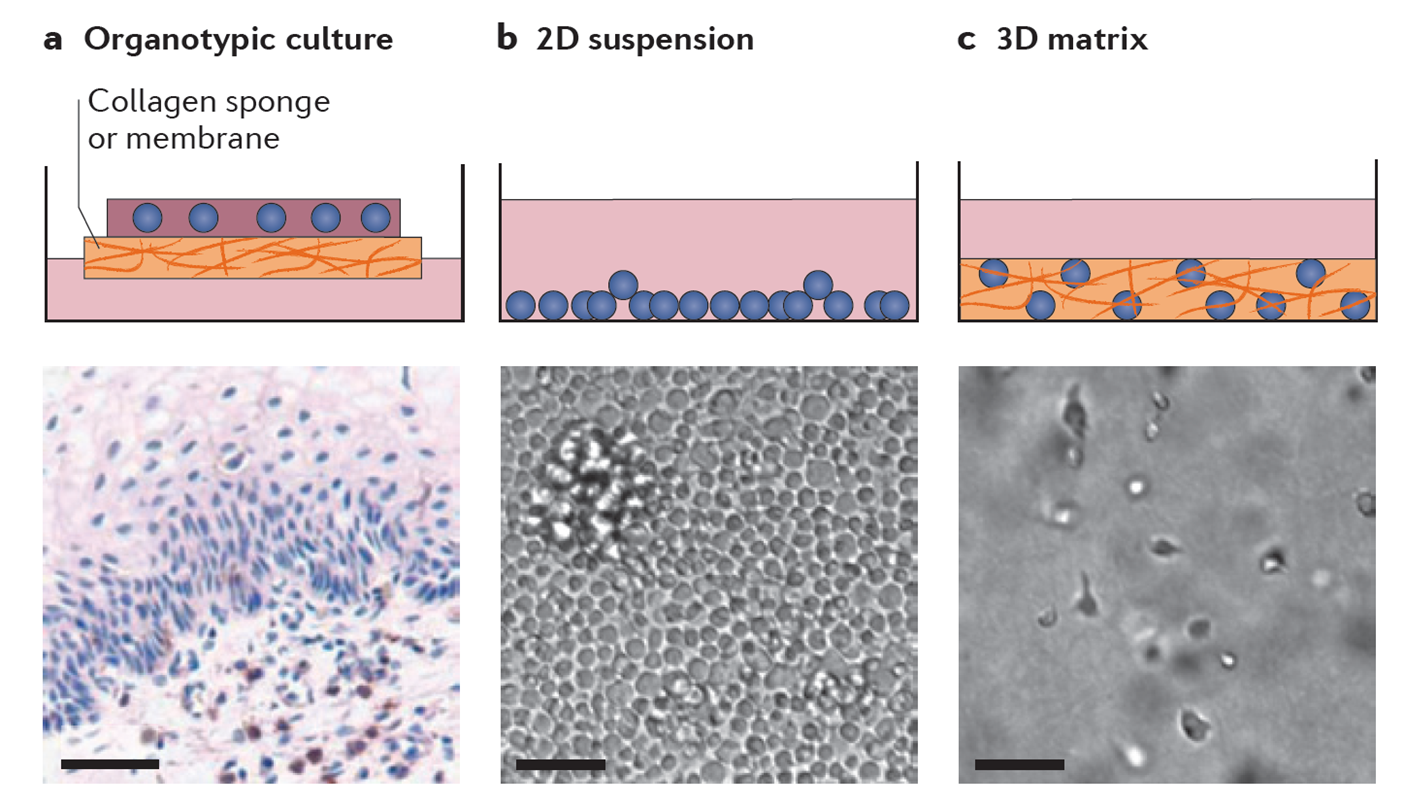
The goal of this project is to develop new ex vivo and in vivo experimental models and study aspects of HIV spread and pathogenesis that are not accessible to experimental manipulation in classical cell culture models. During the first funding period we established (i) ex vivo CD4 T lymphocyte cultures in 3D collagen that allow long-term quantitative assessment of parameters governing HIV spread and (ii) an immunocompetent adoptive transfer mouse model for investigations of HIV evasion strategies from humoral immune responses of the host. To assess the relationship between cell motility and virus spread in the 3D collagen system, a workflow for particle tracking and image processing was developed together with the Rohr and Hamprecht groups.
Quantitative kinetic analyses revealed that the extracellular environment restricts HIV spread by cell-free infection, promotes survival of infected cells and shapes modes and efficacy of HIV spread towards motilitybased, cell-associated virus transfer. Through iterative cycles of experimentation and mathematical modeling, these data allowed the establishment of two mathematical models describing the dynamics of virus replication and cell motility/virus transfer together with the Schwarz and Graw laboratories. Work in the immunocompetent mouse model established that the HIV-1 protein Nef potently suppresses the helper function of CD4 T cells and thereby impairs production of antigen-specific antibodies. Building on the generation of these experimental systems and the key findings made, we will pursue two specific aims in the next funding period.
Specific aim 1 will seek to determine the molecular mechanisms by which the 3D environment shapes cell-associated HIV transmission. By including antigen-presenting cells in the 3D culture system, we will study the relationship between T cell activation and virus spread in 3D, including ultrastructural analyses of cell-cell contacts mediating virus spread and cell activation in collaboration with Kräusslich and Funaya/Schwab.
Specific aim 2 will address which steps of humoral immunity are affected by the viral Nef protein and dissect the underlying molecular mechanisms. Applying advanced microscopy and mathematical modeling to these recently established ex vivo and in vivo model systems will provide new insights into physiological determinants of HIV pathogenesis.
Ex vivo model systems (Reference: Fackler, O.T., Murooka, T.T., Imle, A. and Mempel, T.R. (2014).
Adding new dimensions: Towards an integrative understanding of HIV-1 spread.
Nat. Rev. Microbiol., 12: 563-574)

Figure 7. Schematic model of the disruption of T-cell help by HIV-1 NEF.
Overview of the effects of HIV-1 NEF at the level of the GC (upper panels) and the T-B IS (lower panels).
A Undisturbed physiological reactions in the absence of Nef.
B Effects in the context of HIV-1 infection due to expression of NEF. See text for details.
__________
Kaw, S., Ananth, S., Tsopoulidis, N., Morath, K., Coban, B.M., Hohenberger, R., Bulut, O.C., Klein, F., Stolp, B. and Fackler, O.T. (2020). Expression of HIV-1 pathogenesis factor NEF in CD4 T cells impairs antigen-specific B-cell function. EMBO J e105594.
.
Project staff

.
Samy Sid Ahmed, PhD student

.
Lara Gallucci, Postdoc

